 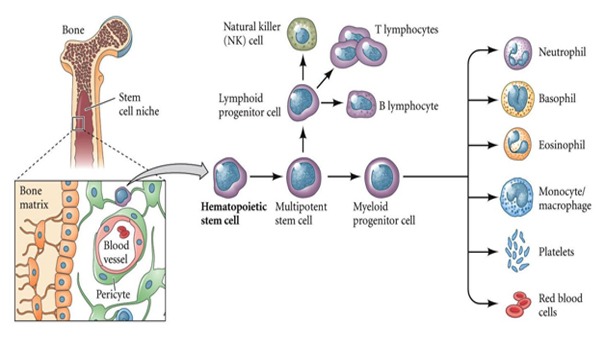
Although there are resident stromal cells in the AGM that support haematopoiesis 31, 32, little is known about their developmental origins, transcriptional and functional identity and contributions to the generation of long-term repopulating HSCs (LT-HSCs).Ī, Schematic outline of experiments performed using E11.5 Nestin-GFP + embryos. Mesenchymal stem cells (MSCs) and their progeny are important constituents of the niche that regulates the size of the HSC pool in adult bone marrow 29, 30. These signals facilitate haematopoiesis in part by regulating the expression of critical haematopoietic transcription factors, including components of the FLI1, GATA2 and SCL transcriptional network, GFI1–GFI1B and RUNX1 (refs. The development of HSCs in the AGM is influenced by NOTCH 19, WNT 20, BMP 21, 22 and other signals 23, 24 from surrounding cells 25. 17) and progresses through pre-HSC stages to generate HSCs in the AGM between E10.5 and E12.5 (ref. Haemogenic endothelium is specified between E8.5 and E10.5 (ref. These HSCs are amplified in the fetal liver 13 and the placenta 15, 16 they take up residence in the bone marrow, which will serve as the major adult site of haematopoiesis. In mouse embryos, the first HSCs appear mid-gestationally (embryonic day 10.5, E10.5) from haemogenic endothelial cells 8, 9, 10 lining the ventral surface of the dorsal aorta through endothelial-to-haematopoietic transition (EHT) 11, 12 in a region known as the aorta–gonad–mesonephros (AGM) 13, 14. The first blood progenitors to emerge (from the extra-embryonic yolk sac) are primitive erythrocytes, followed by erythroid–myeloid progenitors 7. The haematopoietic system in the embryo develops in successive waves 6. Advances in knowledge of embryonic haematopoiesis have informed methods that have been used to produce HSC-like cells in vitro 3, 4, 5. 
As such, understanding the rules that govern HSC emergence, proliferation and maturation is important to reproduce these phenomena in vitro 2. Haematopoietic stem cells (HSCs) have extensive self-renewal capacity and are the source of daughter cells that proliferate, mature and develop into blood cells of all types 1. Together, aorta–gonad–mesonephros Mesp1 der PSCs could potentially be harnessed to manufacture LT-HSCs from endothelium. Dose-dependent inhibition of PDGFRA or BMP, WNT and NOTCH signalling interrupted this reprogramming event. Co-aggregating non-haemogenic endothelial cells with Mesp1 der PSCs but not Wnt1 der PSCs resulted in activation of a haematopoietic transcriptional programme in endothelial cells and generation of LT-HSCs. Here we show that mesoderm-derived PDGFRA + stromal cells ( Mesp1 der PSCs) contribute to the haemogenic endothelium of the dorsal aorta and populate the E10.5–E11.5 aorta–gonad–mesonephros but by E13.5 were replaced by neural-crest-derived PSCs ( Wnt1 der PSCs). We investigated whether mesenchymal stem cells, which provide an essential niche for long-term HSCs (LT-HSCs) in the bone marrow, reside in the aorta–gonad–mesonephros and contribute to the development of the dorsal aorta and endothelial-to-haematopoietic transition. Mouse haematopoietic stem cells (HSCs) first emerge at embryonic day 10.5 (E10.5), on the ventral surface of the dorsal aorta, by endothelial-to-haematopoietic transition. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
